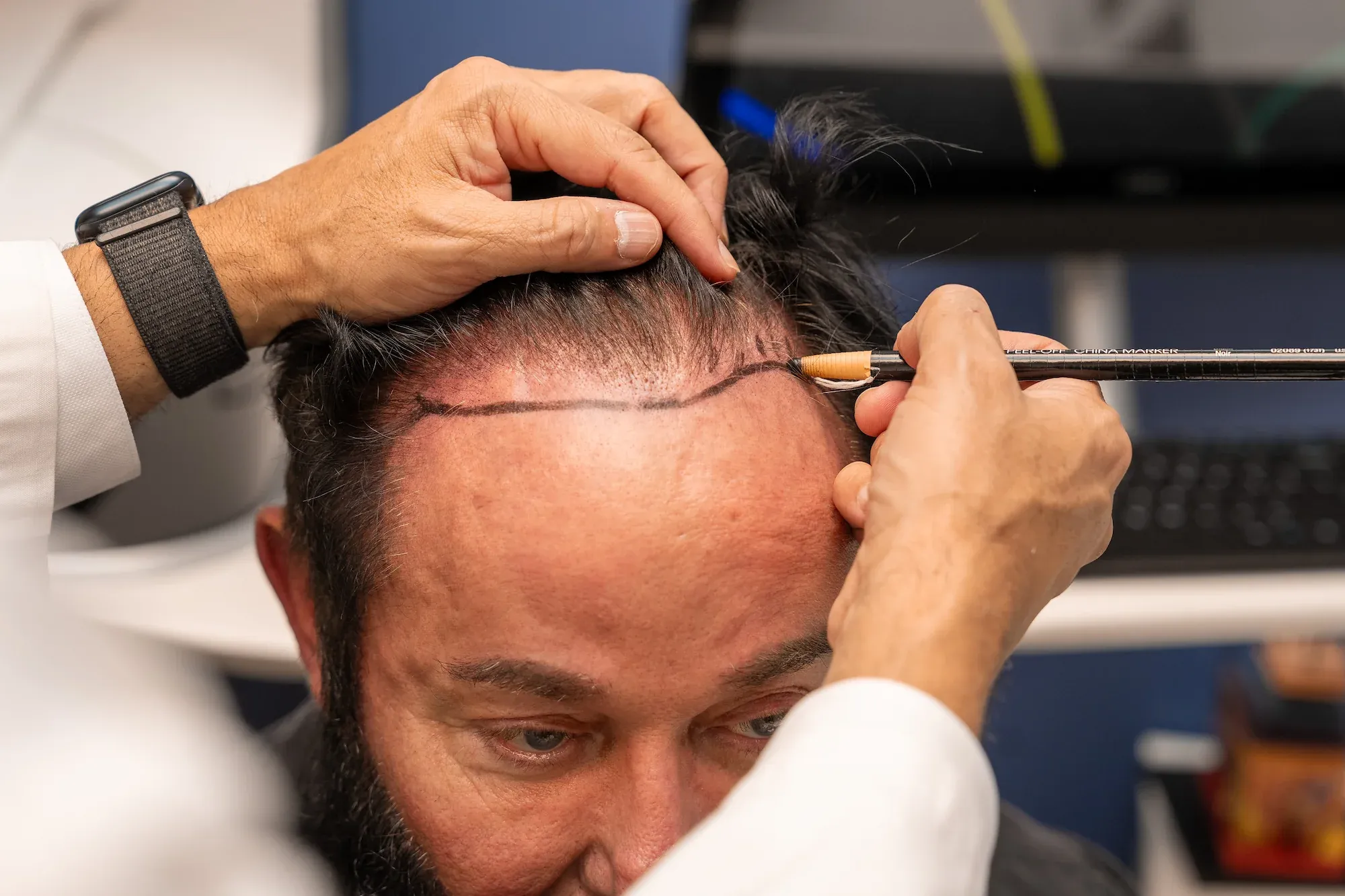
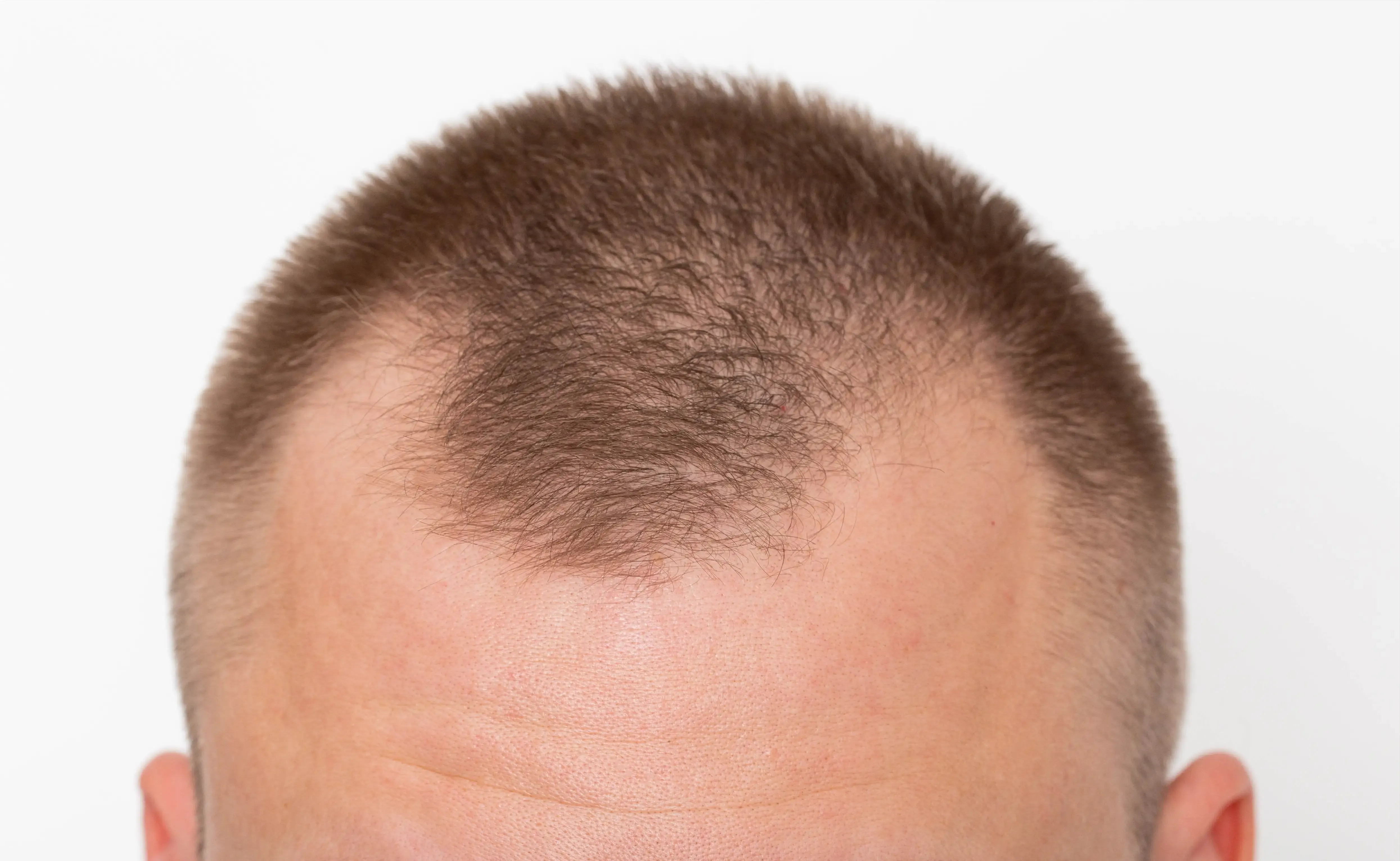
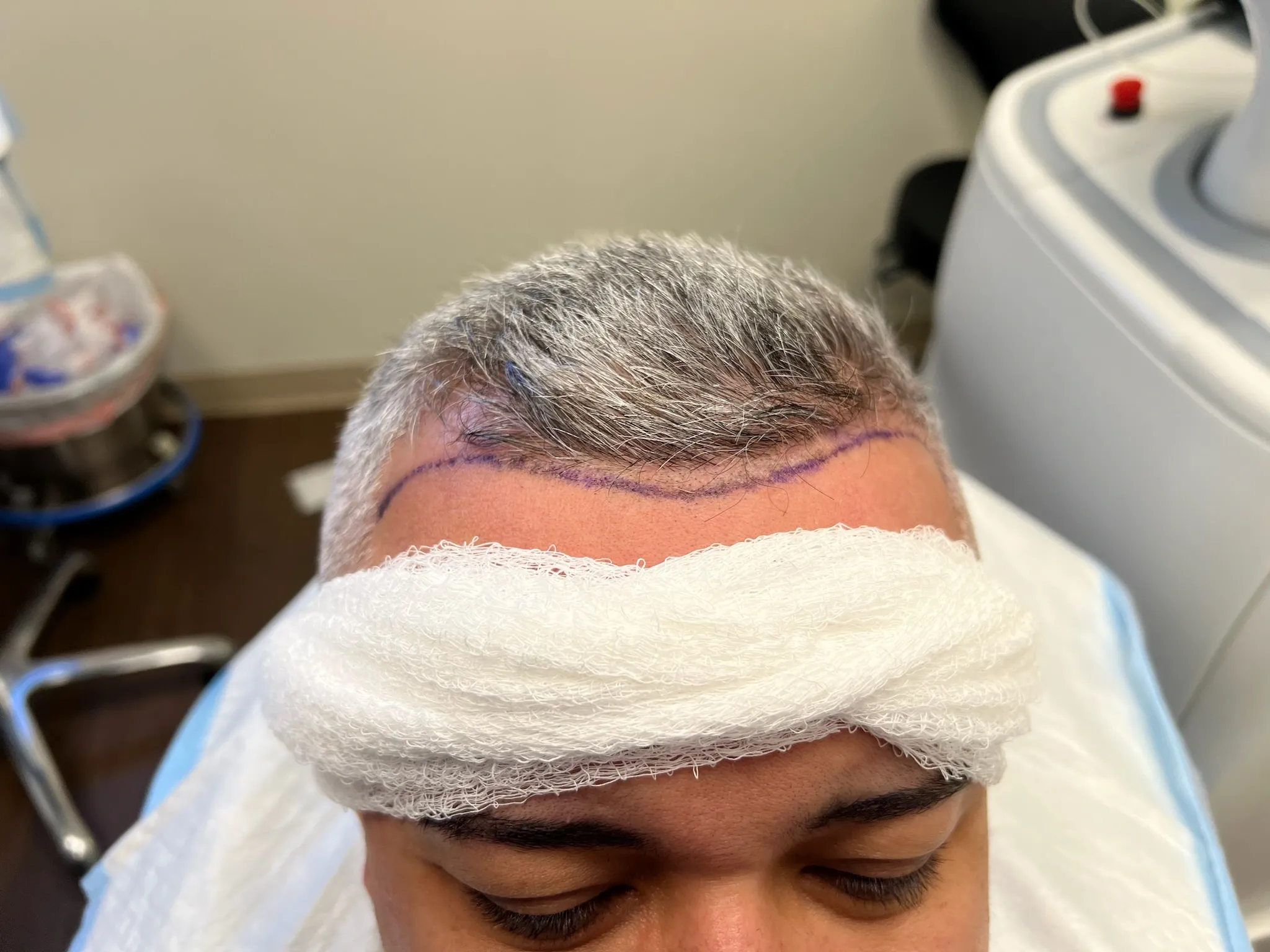
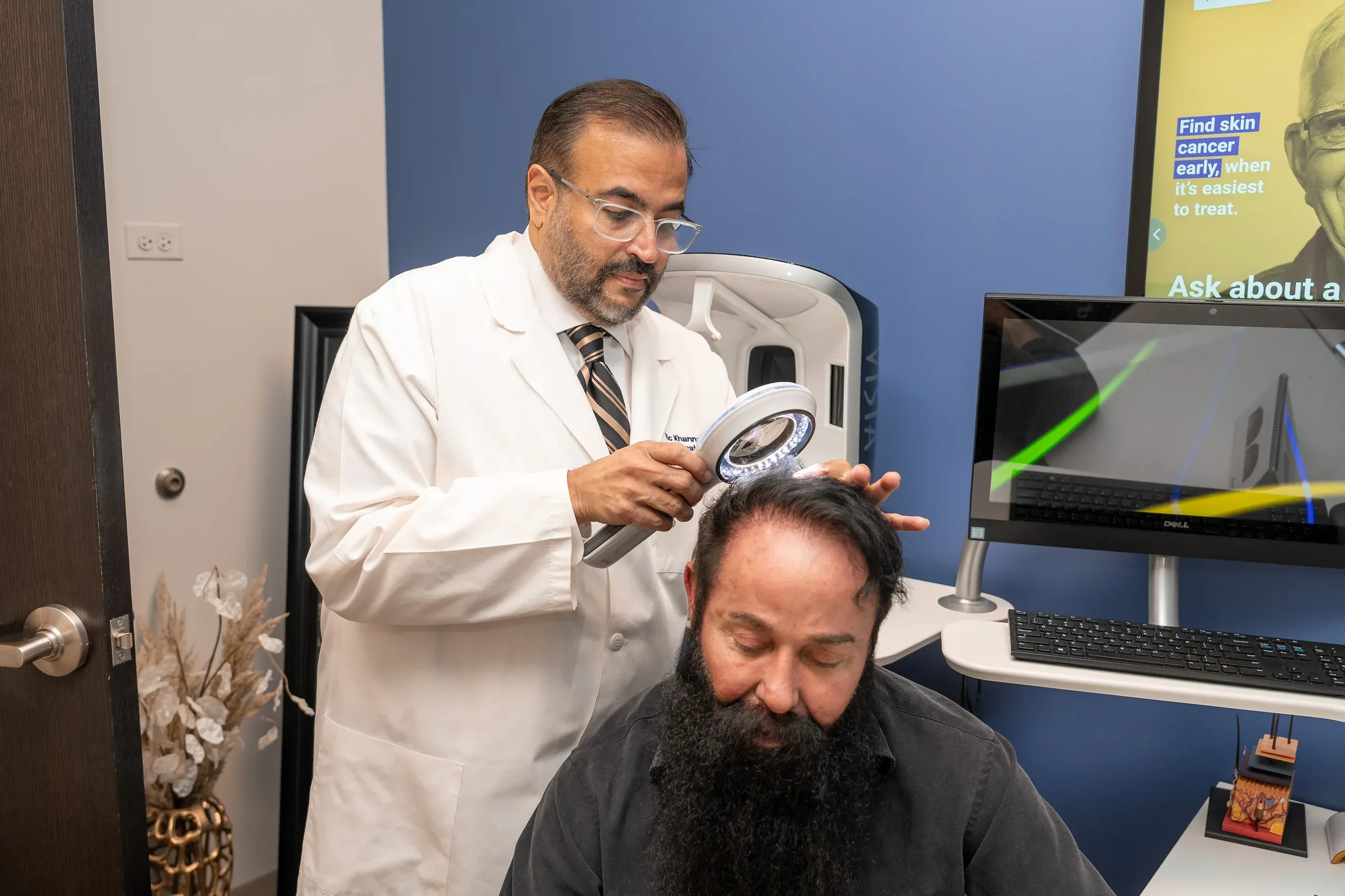
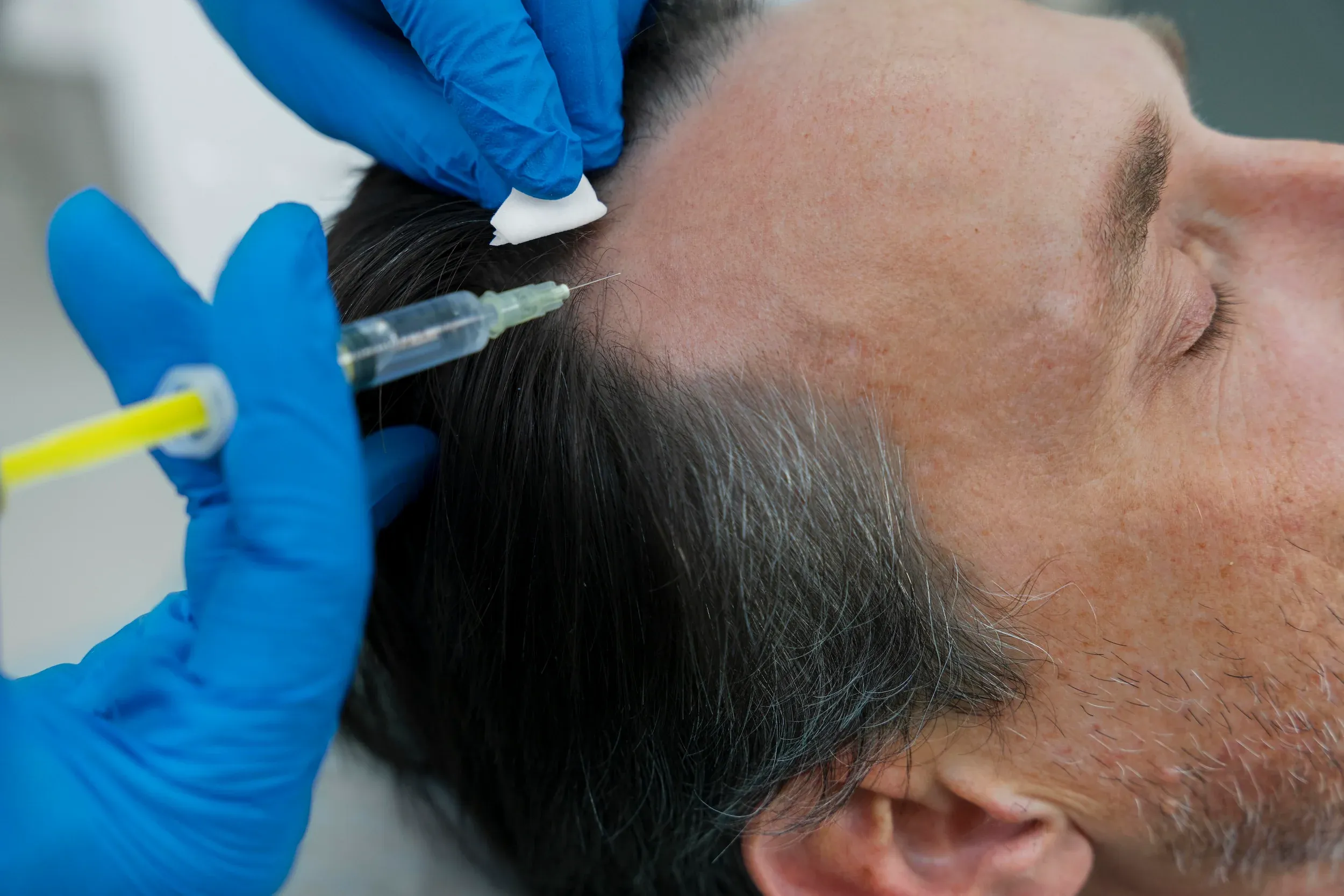
The frontal hairline is uniquely vulnerable because hair follicles in this region contain the highest concentration of androgen receptors. In androgenetic alopecia, dihydrotestosterone (DHT) binds to these receptors and progressively miniaturizes follicles, shrinking terminal hairs into fine vellus hairs over cycles lasting 2–6 years each. Studies show that frontal follicles express up to 40% more 5-alpha reductase—the enzyme that converts testosterone to DHT—than occipital (back-of-head) follicles.
This miniaturization process shortens the anagen (growth) phase from a healthy 3–5 years down to mere weeks, producing hairs too fine and short to provide visible coverage. As each growth cycle completes, the replacement hair emerges thinner and lighter until the follicle eventually ceases producing visible hair altogether.
In frontal fibrosing alopecia, the mechanism is entirely different: an autoimmune inflammatory response targets the hair follicle’s stem cells, causing permanent scarring that destroys the follicle. This condition has increased an estimated 4-fold over the past two decades, particularly affecting women over 50, and often involves eyebrow loss as an early indicator.